My Role
Senior Product Designer (Discovery Lead ) • Log my Care
Timeline: July - October 2025: approx. 3 months
What the project was
Medication management is one of the highest risk workflows in social care. eMAR is not a feature set - it is clinical accountability infrastructure used daily by carers in CQC-regulated UK care settings.
I led the end to end discovery and strategic definition of Log my Care's Electronic Medication Administration Record - a safety critical system used by thousands of carers across the UK to administer, record and audit medication.
Designing for this space required clarity, humility and close collaboration with real carers and care managers. Medication errors in this environment carry direct safeguarding and audit consequences.
The Challenge
As the product had scaled, medication workflows had grown more complex and interdependent than their original design could support. No shared model of the medication lifecycle existed across the organisation. Feature prioritisation was driven by stakeholder opinion rather than the people actually using the system every day.
Every day, carers were managing high risk medications in a system that couldn't be consistently trusted. The cost wasn't just operational. It was human.
The work focused on uncovering the root causes behind:
Medication errors and missed doses
Stock discrepancies and audit gaps
Incomplete or unreliable MAR charts required for CQC compliance
Workflow stress in high pressure care environments
“When we’re dealing with time critical stuff… that can be the difference to hospital admissions, life, death.”
— Care Manager, User Interview 6
What I Did
I mapped the system from the ground up - putting carer voices at the centre of every structural decision made, not internal assumptions or feature requests.
Worked closely with Developers, PMs, Sales, Customer Success
Conducted and synthesised 14 user interviews across carers, managers and support staff. Interview recordings captured and organised in Dovetail.
Ran 9 internal stakeholder interviews (Across Product, Engineering, Sales, Customer Success, CTO and CEO) to identify patterns, themes, gather constraints and align understanding
Gathered 69 survey responses from existing and churned users
Proactively reached out to both churned and existing users, building rapport and trust to encourage participation.
Mapped the full end to end medication lifecycle (Adding, Administering, Updating/Pausing, Stock, Exceptions)
Benchmarked leading eMAR competitors to assess structural gaps and differentiation opportunities
Facilitated alignment with PM, CTO and Engineering
Presented findings and design solutioning plan to the full company in an in office sessions
Defined problem statements, JTBD and success criteria adopted across Product and Engineering.
Interview recordings captured and organised in Dovetail as part of the 14-participant discovery phase. These sessions represented carers, managers and support staff across a range of care settings.
What I Found
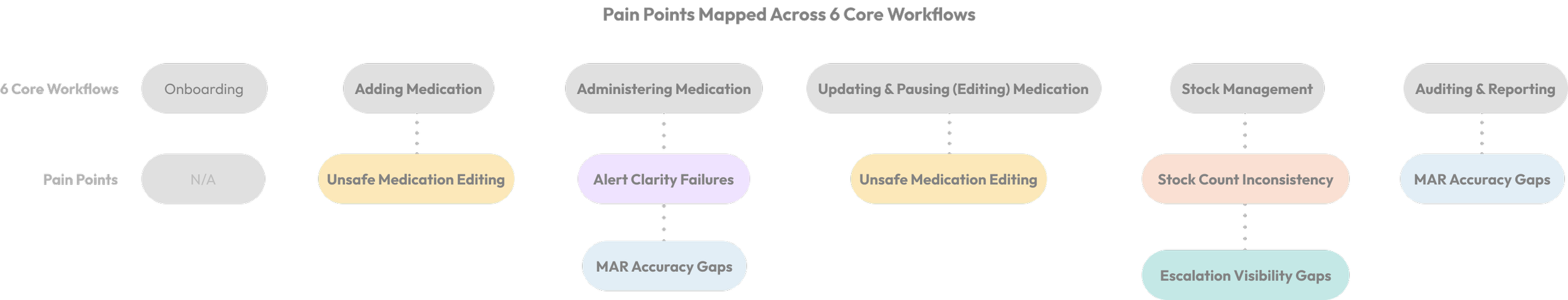
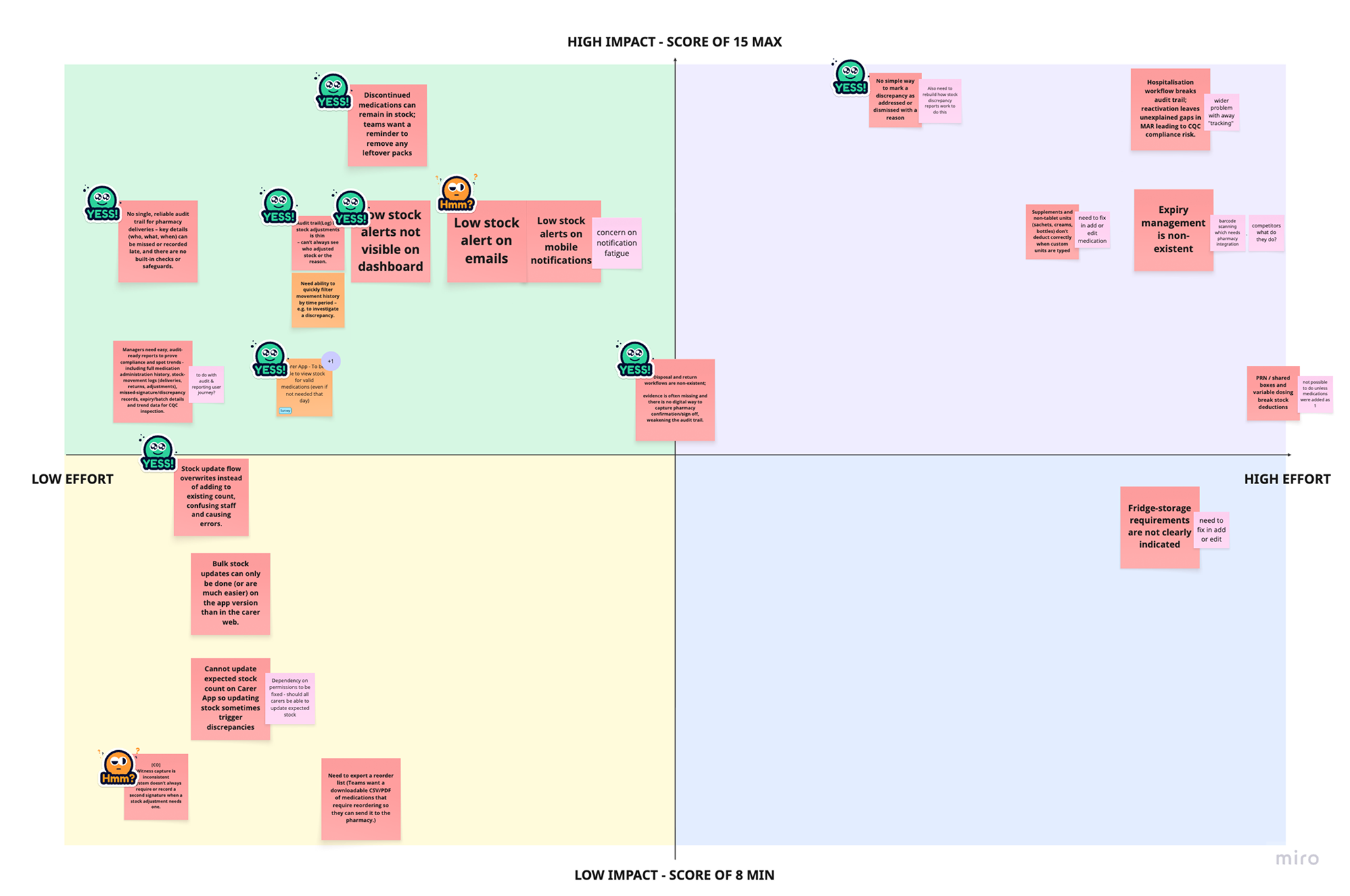
What emerged wasn't a list of UI complaints. It was a system with compounding failure modes across six interdependent workflows - where a failure in one stage made the next stage worse. Critically, many of these risks were previously invisible - stock logic gaps, inconsistent audit trails and missing escalation paths had been contributing to errors and support load without being formally recognised or connected.
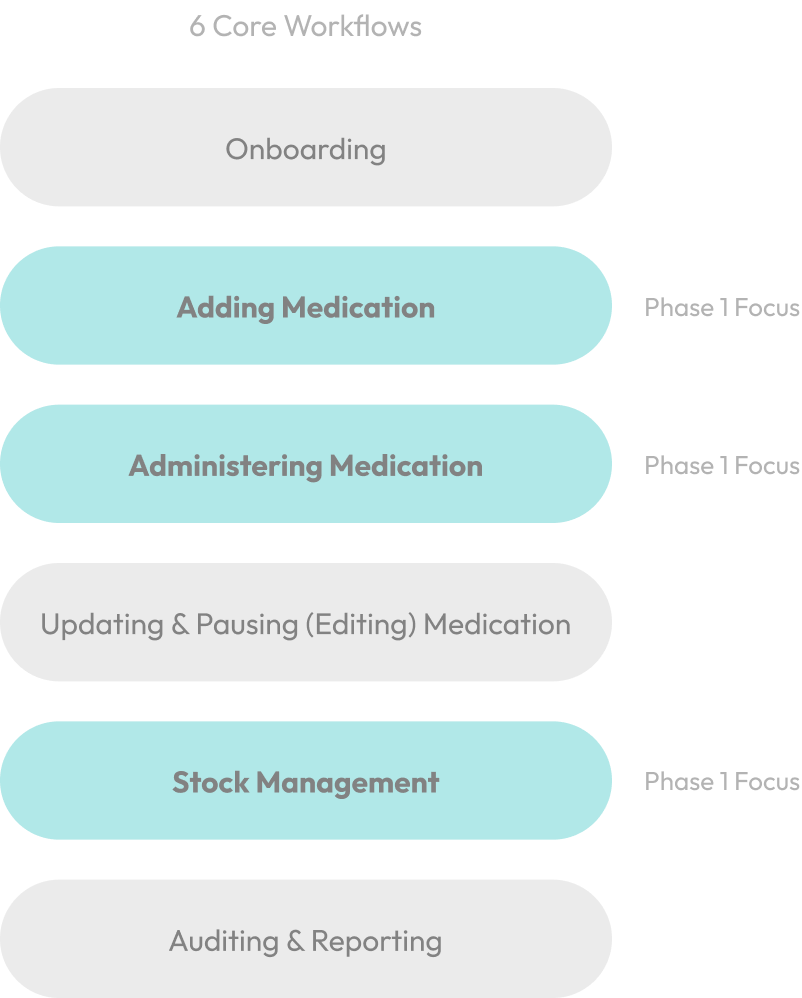
6 interdependent workflows mapped end to end for the first time:
Onboarding
Adding Medication
Administering Medication
Updating & Pausing (Editing) Medication
Stock Management
Auditing & Reporting
Initial deep risk analysis focused on Adding, Administering and Stock Management due to highest frequency and safety exposure.
Full walkthrough available on request during interviews.
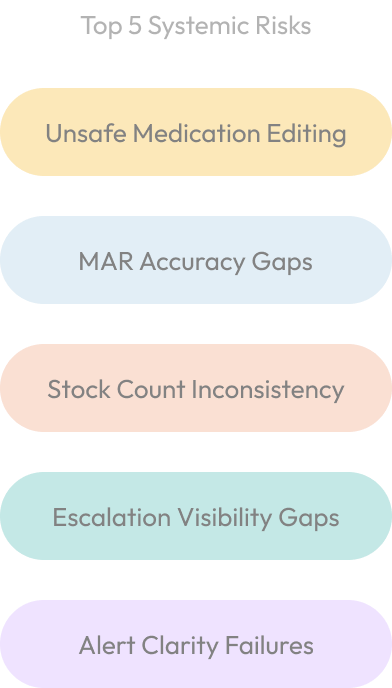
Five cross workflow safety failure patterns from 200+ synthesised insights:
Unsafe Medication Editing
Inability to safely edit or update medications
MAR Accuracy Gaps
Missing or inaccurate MAR charts
Stock Count Inconsistency
Stock inconsistencies and audit gaps
Escalation Visibility Gaps
No clear way to flag issues
Alert Clarity Failures
Overwhelming, unclear, or missing alerts
The critical insight: unsafe editing in Adding directly created MAR gaps in Administering. Stock inconsistencies created escalation blind spots in Auditing. These weren't parallel problems. They were connected.
These failures appeared consistently across 14 interviews and were reflected in support tickets and usage trends.
What I Decided
I introduced a risk based prioritisation model combining frequency, safety exposure and operational impact - replacing opinion led roadmap debate with evidence led sequencing.
Not every decision was popular. I recommended descoping two high visibility features from Phase 1 because neither addressed any of the 5 structural failure patterns. Fixing the surface without fixing the system underneath would have created false confidence in a still broken record.
I also established a safety led operating model with explicit decision gates between discovery and build. When engineering pushed to ship ahead of validation I held the sequencing. In a CQC regulated product, shipping first and validating later isn't a velocity decision. It's a safety risk.
My safety led operating model
What Changed
At the close of discovery I presented findings to the full company - not as a research readout, but as a deliberate attempt to create shared understanding across teams who had never spoken directly to carers or care managers.
The conversation shifted visibly:
Before:
"Which features should we prioritise?"
After:
"What do we need to understand before we redesign this?"
That shift created the organisational conditions for the right redesign to happen. This work didn't just shape the eMAR redesign - it reshaped how the organisation approaches problem solving and clinical safety.
What I established:
A unified system model across 6 interdependent workflows - aligning PMs, sales, customer service, developers and leadership around shared failures and user needs for the first time (This didn’t exist before I joined)
Leadership aligned on highest risk structural failures before a single design decision was made
Outlined initial workflow directions for all medication tasks
Defined problem statements, opportunity areas, ‘Jobs to be done’ (JTBD) and success criteria adopted across Product and Engineering - creating a shared language for what carers needed to accomplish, not just what they were complaining about
Evidence based prioritisation that directly shaped Phase 1 scope
Explicit safety decision gates reducing ambiguity between discovery and delivery
An 11 step design solutioning framework preserving safety critical insights through to build
Produced a prioritised pain point model (mentioned in X/14 interviews) giving teams a single evidence based understanding of what was truly affecting medication safety
Created a concise cross team report and documentation that replaced scattered knowledge with a stable reference for roadmap planning
Prioritisation Model
I developed the 11 step solutioning plan to ensure the redesign phase had clear structure, alignment and expectations across design, product managers, development, sales and customer success.
Presented the findings and the solutioning plan to the full company in an in office session to ensure cross team visibility and buy in.
Solution foundations for the redesign
I left the organisation before the full UI redesign phase. The work below represents the conceptual foundations the team used to begin solutioning.
A clearer, safer Administering Medications structure
step based clarity, stronger dose checking, reduced cognitive load during rounds, safe error handling and missed dose patterns
A modernised Stock Management model
ideal relationship between stock, MAR entries and exceptions, simple guided top up flows
A scalable interaction model across medication tasks
modular patterns that future features could plug into
Solution principles to guide future UI
Safety first. Clear timely visibility of critical alerts. Reduce cognitive load. Clarity over density. Predictable consistent patterns across all tasks.
What I learned
The biggest tension in regulated discovery is pace. Leadership wanted solutions before the system model was stable. Safety critical framing only lands when you give leadership a risk cost for moving too fast - not just a design rationale. I'd quantify the regulatory exposure earlier next time to accelerate alignment.
Safety must be architected before it is designed. That principle shaped every decision I made on this project.