DESIGNING AT LOG MY CARE
UI/UX Design · Discovery & Research · Mentoring & Design Leadership • Systems Thinking · Stakeholder Alignment · Safety Critical Design · User Interviews · Synthesis & Prioritisation · Regulated HealthTech · Figma
DESIGNING AT LOG MY CARE
UI/UX Design · Discovery & Research · Mentoring & Design Leadership • Systems Thinking · Stakeholder Alignment · Safety Critical Design · User Interviews · Synthesis & Prioritisation · Regulated HealthTech · Figma
“Care management software for next generation care providers”
Working at Log my CARE
Log my Care is a UK based care management platform used by thousands of carers and care managers in CQC-regulated settings across the country. Built to support adult social care providers, it handles some of the most safety critical workflows in the sector - including medication management, daily care notes and compliance reporting.
As Senior Product Designer, I was brought in to lead discovery on their eMAR (Electronic Medication Administration Record) system - the medication management workflow at the heart of daily care delivery. Working across product, engineering, sales, customer success and clinical stakeholders, I mapped the full medication lifecycle, surfaced systemic failure patterns and established the strategic foundations for a safer, more compliant redesign. I also mentored two designers during the engagement, supporting them through research synthesis and design critique.
Key Contributions:
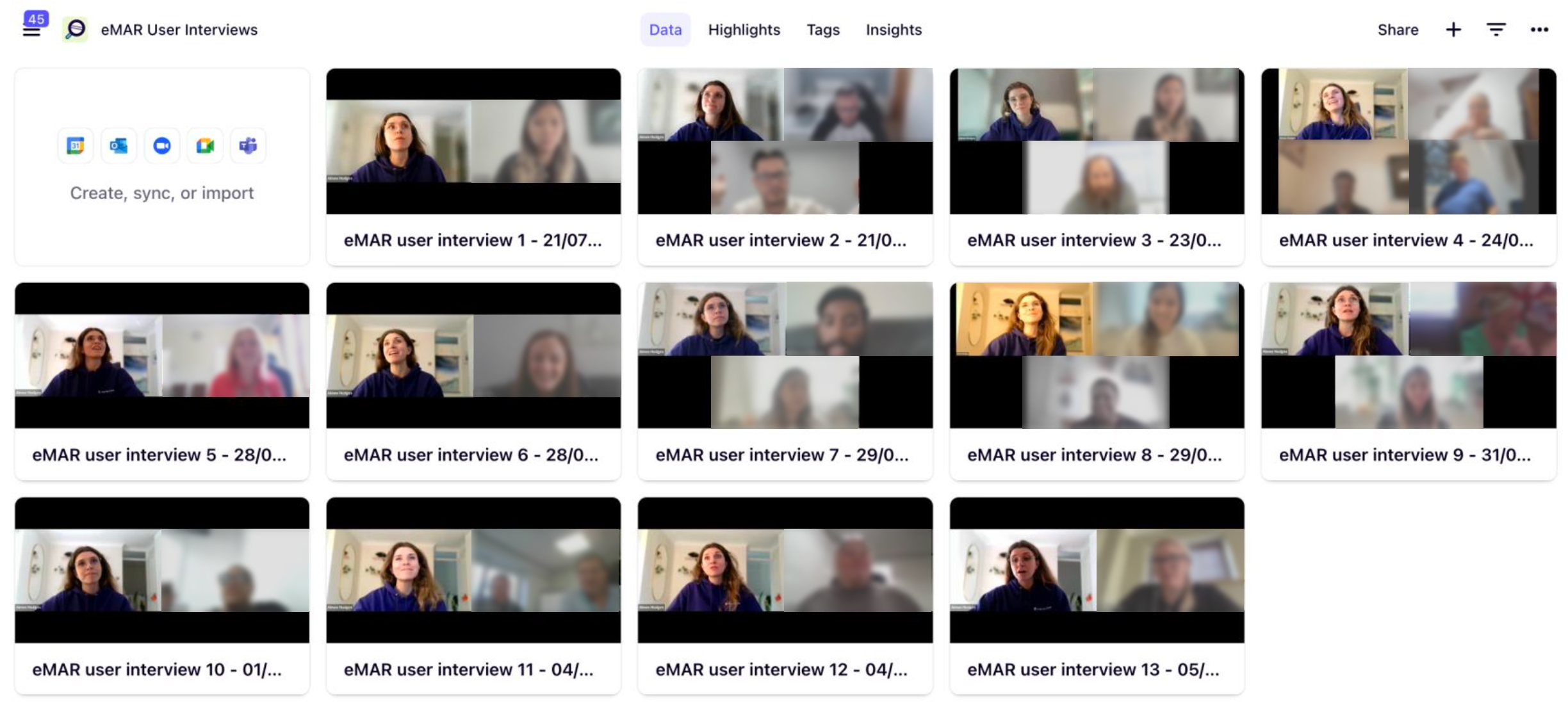
eMAR Discovery Leadership: Led end to end discovery across 6 interdependent medication workflows, conducting 14 user interviews, 9 stakeholder sessions and 69 survey responses, synthesising 200+ insights to surface 5 systemic safety failure patterns.
Systems & Workflow Mapping: Built a complete model of the medication lifecycle where none existed, creating shared organisational understanding across product, engineering, sales, customer success and clinical stakeholders for the first time.
Strategic Influence & Prioritisation: Introduced evidence based prioritisation to replace opinion led decision making, directly shaping the scope and sequencing of the redesign before a single UI decision was made.
Risk Identification: Caught a critical permissions conflict during discovery that would have created significant downstream rework across engineering - flagged and resolved before it became a problem.
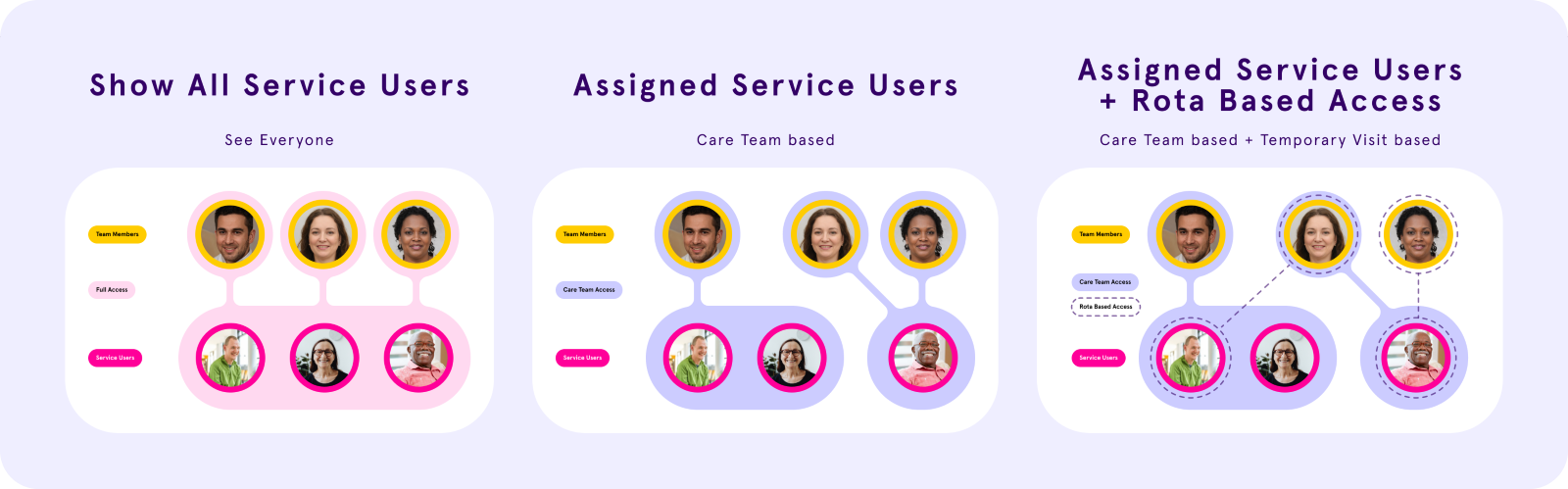
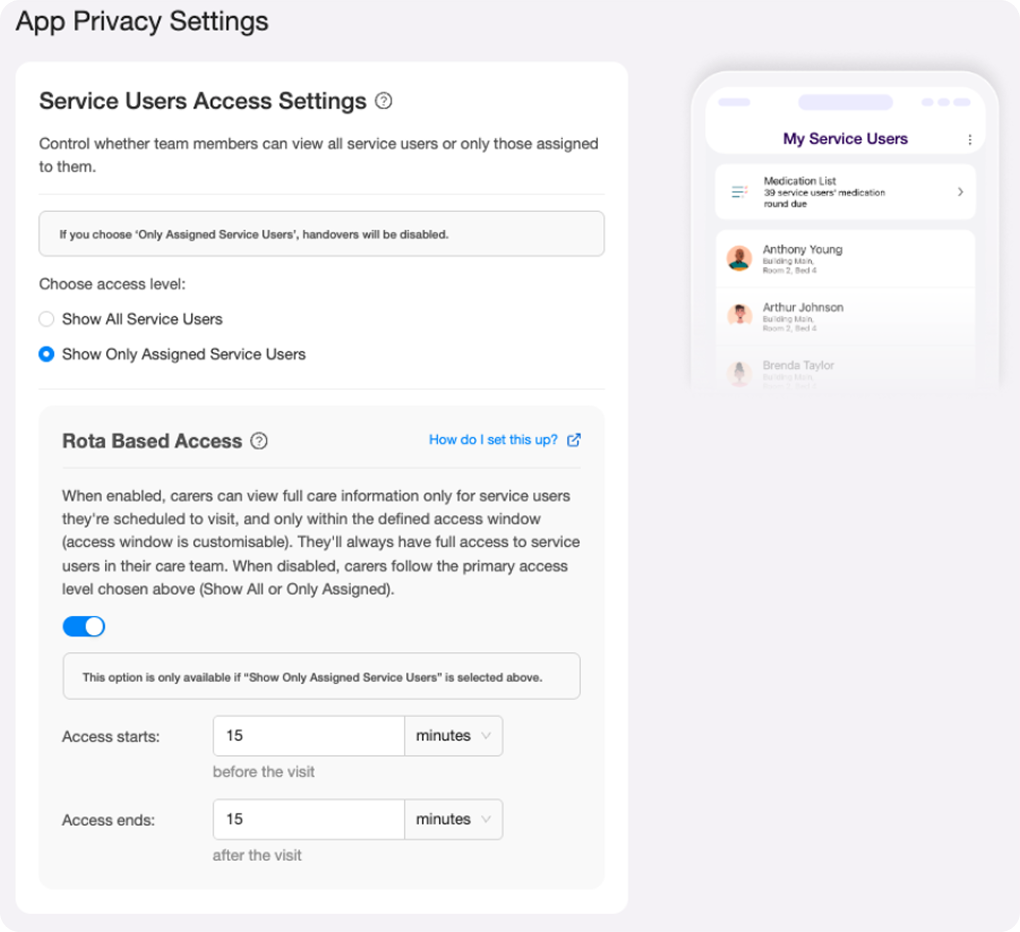
Rota-Based Access Controls: Designed a privacy first access control system end to end, from concept through interaction design and UI copy - four access states, time-based visit windows, and a settings interface built for non technical care managers.
Regulated Domain Expertise: Designed across CQC regulated workflows where errors carry real clinical and safeguarding consequences, bringing appropriate rigour to research, synthesis and design decisions throughout.
Design Mentoring: Supported two designers during the engagement, providing hands on guidance through research synthesis, insight framing and design critique.
De-Risking Medication Management Through eMAR Discovery
End to end discovery to define a safer, CQC compliant medication management system for UK care providers.
Discovery Lead · eMAR · User Research & Insight · Safety Critical Design · Systems Thinking · Stakeholder Alignment · Evidence Based Prioritisation · Regulated HealthTech · CQC Compliance · Log my Care
De-Risking Medication Management Through eMAR Discovery
End to end discovery to define a safer, CQC compliant medication management system for UK care providers.
Discovery Lead · eMAR · User Research & Insight · Safety Critical Design · Systems Thinking · Stakeholder Alignment · Evidence Based Prioritisation · Regulated HealthTech · CQC Compliance · Log my Care
Interview recordings captured and organised in Dovetail as part of the 14-participant discovery phase. These sessions represented carers, managers and support staff across a range of care settings.
My Role
Senior Product Designer (Discovery Lead ) • Log my Care
Timeline: July - October 2025: approx. 3 months
Medication management is one of the highest risk workflows in social care. eMAR is not a feature set - it is clinical accountability infrastructure used daily by carers in CQC-regulated UK care settings.
As Log my Care's eMAR had scaled, the complexity of medication workflows had outgrown the system's original design. No shared model of the medication lifecycle existed across the organisation.
I led end to end discovery to build the structural foundation the redesign needed to be safe. 14 user interviews, 9 stakeholder sessions, 69 survey responses and 200+ synthesised insights later, five cross workflow safety failure patterns emerged that reshaped how the organisation approached the problem entirely.
Designing for this space required clarity, humility and close collaboration with real carers and care managers. This work didn't just shape the eMAR redesign. It reshaped how the organisation approaches problem solving and clinical safety.
Want to see the full case study?
The full case study covers the complete discovery arc: system mapping, failure pattern analysis, prioritisation decisions, leadership challenges and solution foundations.
Rota-Based Access Controls, Built Around How Homecare Actually Works
Flexible, privacy first access control designed around how homecare providers actually staff their teams.
Systems Thinking · Interaction Design · UI Copy · B2B SaaS · Data Privacy · Access Control · Regulated HealthTech • Log my Care
Rota-Based Access Controls, Built Around How Homecare Actually Works
Flexible, privacy first access control designed around how homecare providers actually staff their teams.
Systems Thinking · Interaction Design · UI Copy · B2B SaaS · Data Privacy · Access Control · Regulated HealthTech • Log my Care
My Role
Senior Product Designer • Log my Care
Timeline: May - July 2025: approx. 2 months
Data privacy in care isn't a nice to have. It's a safeguarding requirement - and the system carers use every day had no way to enforce it properly.
I designed a rota-based access control system for Log my Care, giving homecare providers precise control over which carers can see sensitive service user information and when. In a sector that relies heavily on agency and temporary staff, the existing binary toggle wasn't enough. Four access states, time based visit windows, and a settings UI built for non technical care managers - designed end to end.